Reimagining
drug discovery
Standard approaches to drug discovery have historically taken a limited view, using technology that identifies, for example, only genetic mutations or protein overexpression as drug targets. Studying cells in this narrow context provides a one-dimensional view and overlooks other anomalies which may drive disease, for instance, proteins mislocalized in diseased versus healthy cells.
At ZielBio, we see a more complete picture of the disease state. We are ushering in a new era of drug discovery, in which we study cells in their native context, enabling the identification of drug targets previously overlooked by traditional genomic and proteomic strategies. Data that may be lost with other techniques can be preserved and leveraged with our proprietary platform.
Our first target:
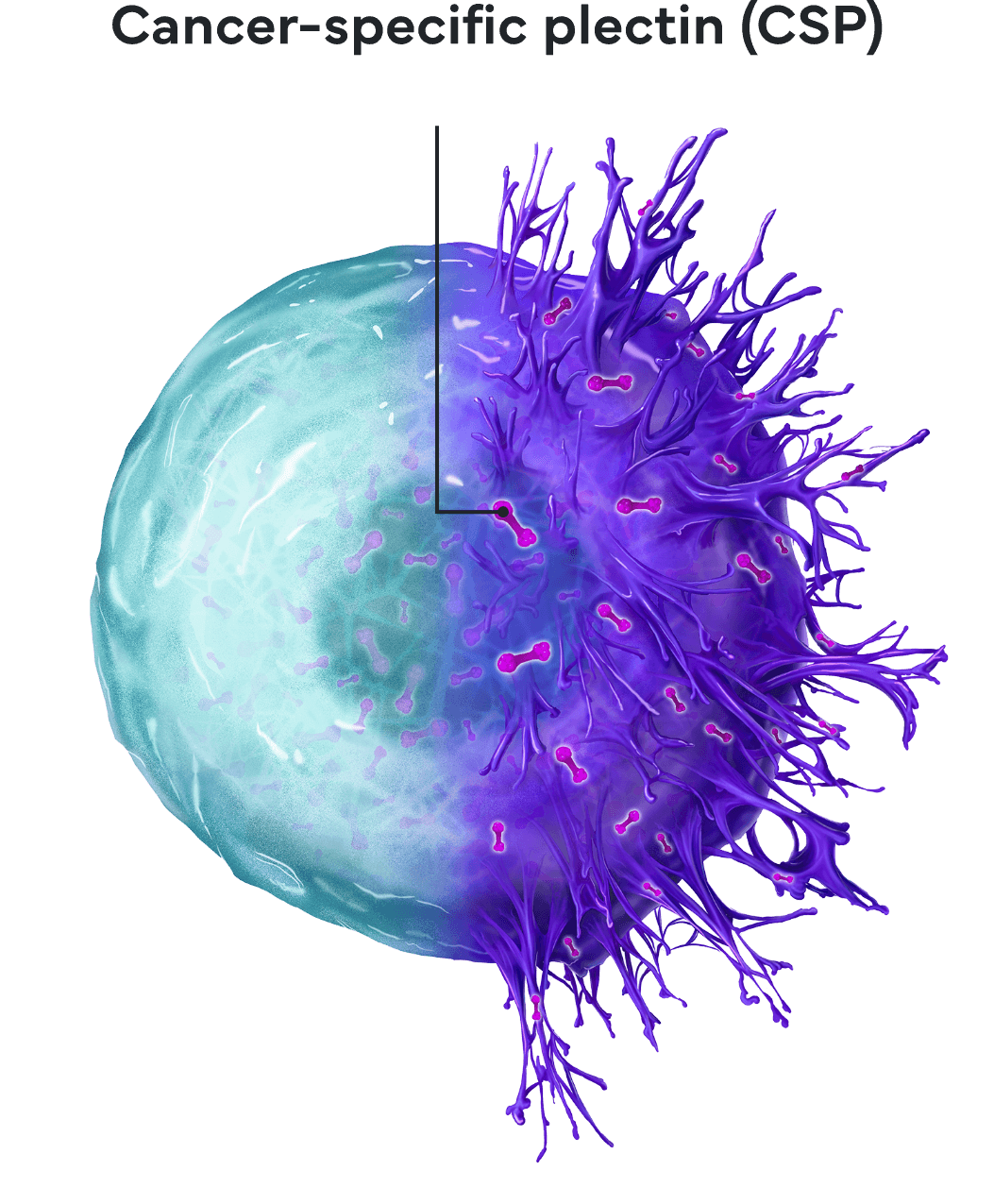
cancer-specific plectin

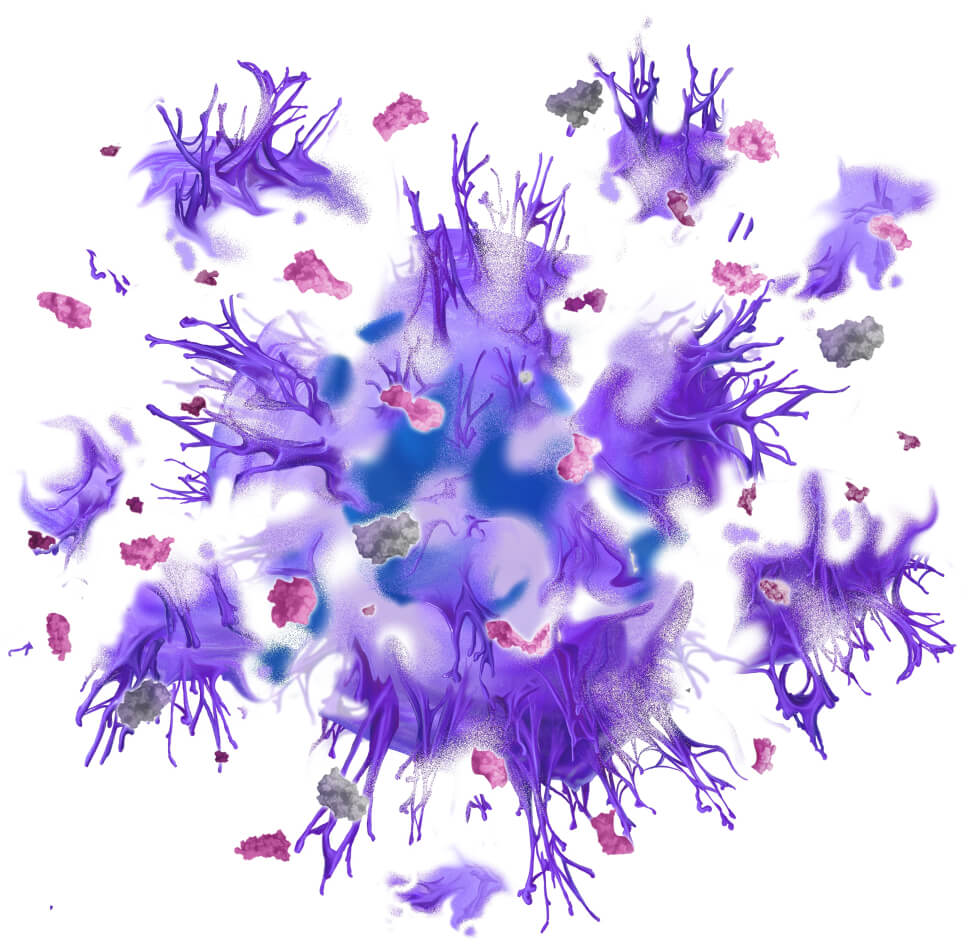
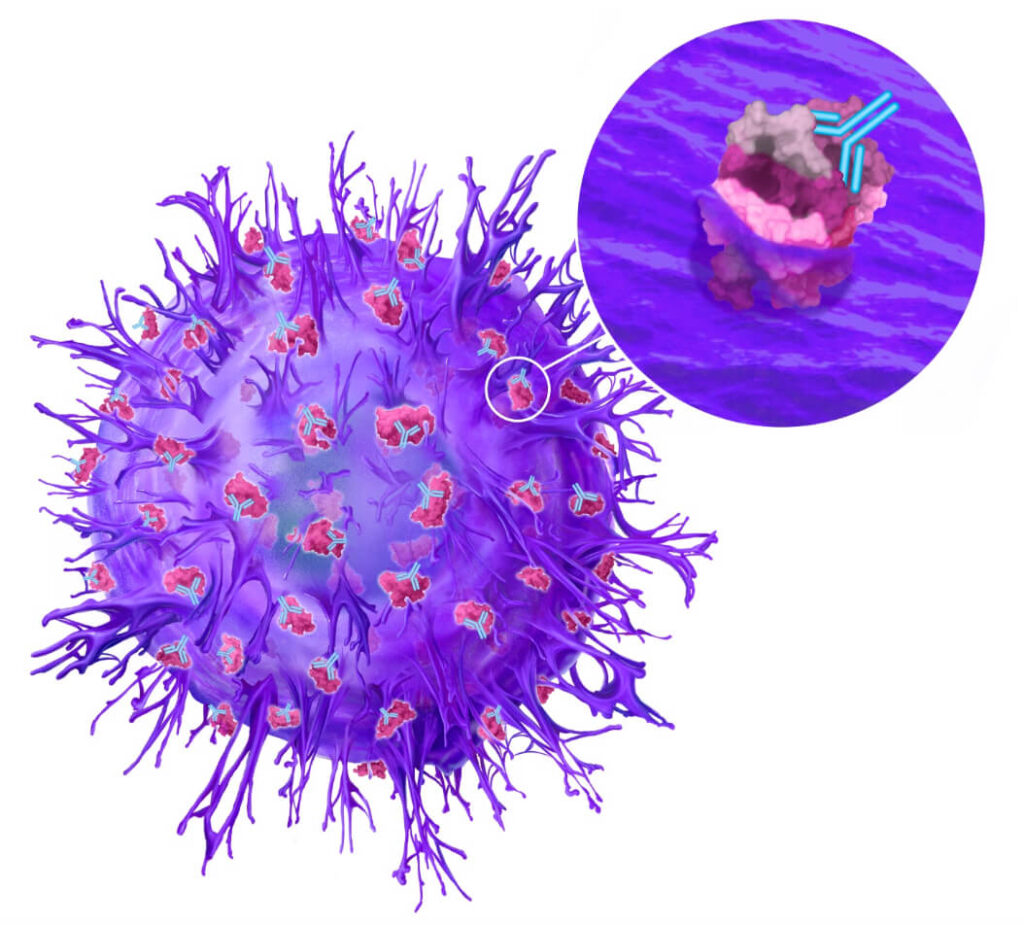
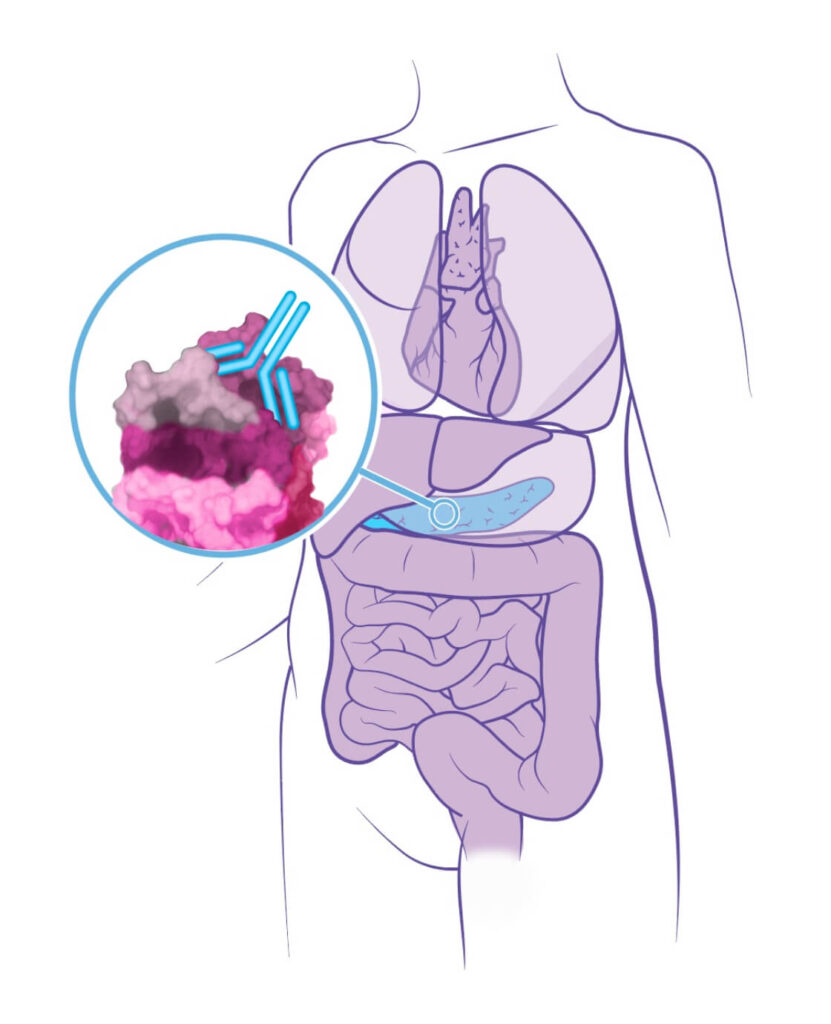
Using our drug discovery platform, we identified and validated our first target: cancer-specific plectin (CSP). While the protein plectin is normally located inside healthy cells, we revealed that in cancer cells, plectin presents on the cell surface, where it is associated with more aggressive disease. CSP is found on numerous cancer types, including several with poor prognosis, such as pancreatic, ovarian, and bile duct cancers. In our extensive preclinical research, our first drug, ZB131, has demonstrated an ability to bind specifically to CSP, resulting in remarkable anti-cancer activity in preclinical models.
We have preliminary clinical data from a Ph 1 study with our anti-CSP monoclonal antibody, ZB131, demonstrating safety and tolerability in a cohort of patients with a variety of solid tumors. We are currently optimizing the use of ZB131 and developing several oncology therapeutics leveraging our knowledge of this target and this antibody. We are also developing a pipeline of novel therapeutics that correspond to several other disease targets beyond CSP.